Amber Baele
Scientist Associate, Flow Cytometry
As we open 2026 with our first Flow Matters blog post of the year, it is the perfect moment to look ahead. Flow cytometry continues to evolve at a rapid pace, driven by technological innovation, expanding clinical applications, and growing expectations around data quality, speed, and standardization. Across research, clinical development, and regulated environments, the field is becoming more complex and more central to decision‑making than ever before. It’s a lot to keep up with. But maybe that’s exactly what makes this technology so exciting.
Below, we outline several key trends shaping the future of flow cytometry.
Why high-dimensional flow cytometry? Because cells have more to say than we ever expected
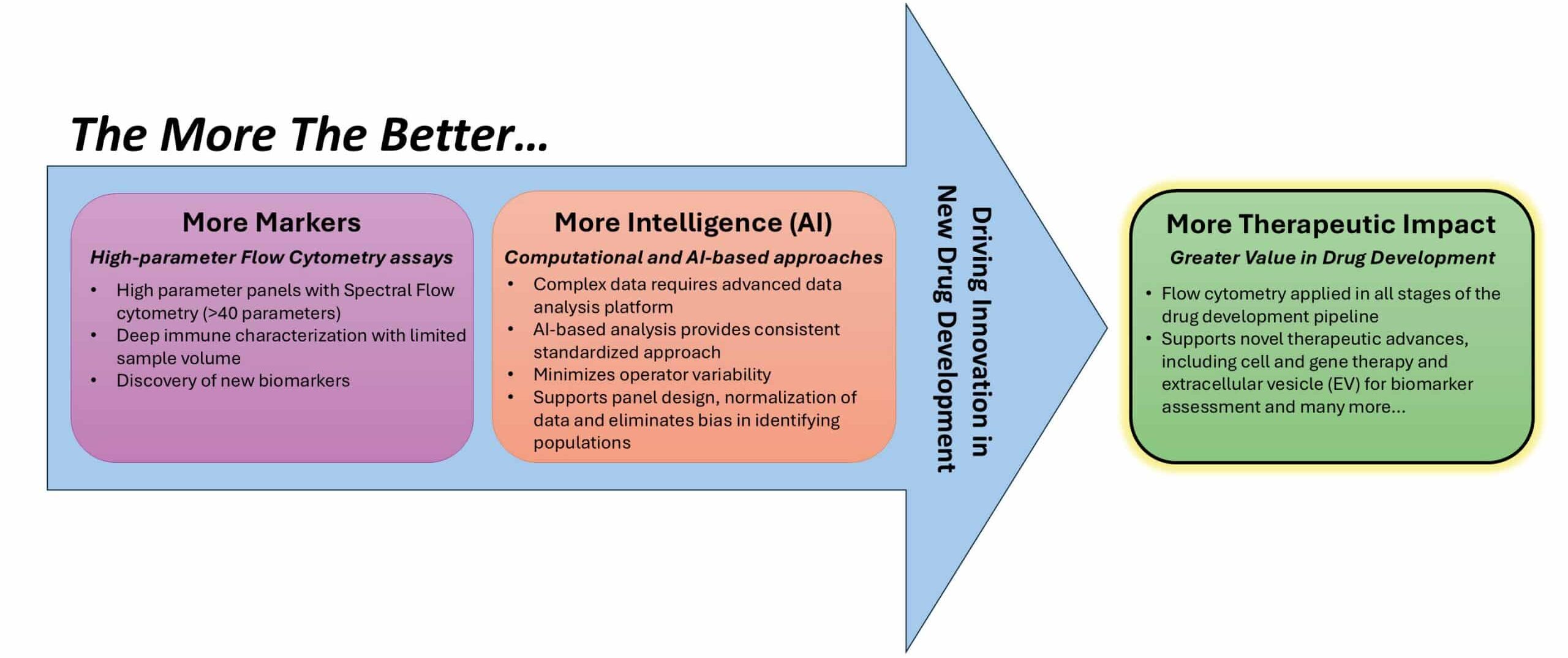
One of the most visible trends in flow cytometry is the continued rise of high‑parameter assays. Conventional flow cytometry, once limited to a small number of markers, has evolved dramatically with advanced technology, such as spectral flow cytometers. Spectral flow cytometers can measure more than 40 parameters, offering exceptional insight into cellular heterogeneity and immune complexity1. Simplicity was never in the cell, only in our tools (instruments). Now that the cell can finally “speak,” the question becomes: how do we learn to “listen”?
Spectral flow cytometry has been a major driver of this shift. By capturing the full emission spectrum of fluorochromes rather than discrete wavelength bands, spectral systems allow researchers to push beyond traditional limits (refer to this blog post for a brief overview of conventional vs. spectral flow cytometers). This enables deeper phenotyping, greater flexibility in panel design, and more comprehensive immune profiling. These capabilities are especially valuable when working with limited sample volume such as BMA, CSF, and blood from pediatric patients. Importantly, these advances do not only apply to classical immune cells, but they also extend our ability to interrogate small particles, such as platelets and extracellular vesicles (EVs), which were previously a challenge to fully characterize.
However, increased dimensionality also introduces new hurdles. High‑parameter panels require advanced expertise in panel design, fluorochrome selection, instrument setup, and validation. Without careful optimization and robust controls, assay complexity can lead to increased variability and challenges in data interpretation. As a result, the value of high‑parameter flow cytometry depends not only on instrumentation but also on experienced scientific oversight.
Why are computational and AI-based data analyses essential in flow cytometry? Because even experts appreciate a second opinion
As the number of measured parameters increases, so does data complexity. How do we handle this? What do we actually do with all of these data? High-dimensional datasets don’t just challenge our hands-on skills; they challenge our brains. Manual gating approaches alone can’t keep up with this exponential increase in markers in a single tube. The current approach is time-consuming, difficult to scale, and subject to analyst-dependent variability.
Artificial intelligence (AI) and machine learning (ML), together with advanced computational algorithms, are increasingly positioned as solutions to these challenges. In clinical flow cytometry, computational and AI‑based data analysis can support multiple steps of the analytical workflow, including data preprocessing, quality control, population identification, and result interpretation. Computational models can assist with automated quality checks, detection of technical artifacts, and harmonization of data across batches or instruments. Unsupervised and supervised learning approaches can identify cell populations, reveal subtle phenotypic shifts, and support data analysis that would be difficult to achieve with manual analysis. By applying objective and consistent criteria for population identification, AI-assisted approaches reduce subjectivity and improve reproducibility across analysts and institutions.
AI can also contribute to panel optimization and identification of suboptimal marker combinations, which further strengthens assay robustness. In this way, AI not only improves data analysis but also reinforces the entire experimental lifecycle by helping to create a stronger flow cytometry panel.
However, technical capability alone is not sufficient in clinical settings. Successful implementation requires careful consideration of regulatory requirements in clinical laboratories. Clinical flow cytometry operates in regulated environments where transparency, traceability, and reproducibility are essential. AI models must therefore be interpretable, validated, and thoroughly documented. Laboratories must understand how populations are defined, how algorithmic adjustments are applied, and how outputs can be traced back to the underlying raw data. Some of the companies developing these tools have already started to implement regulatory requirements, such as 21 CFR Part 11 compliance and GCLP (good clinical laboratory practice).
In clinical labs, AI-driven data analysis could be most effective when integrated into hybrid workflows. Scientists define the questions, validate outputs, and interpret results. AI amplifies our capabilities, but it’s our judgment that gives the results meaning. Without that, we risk insights that are technically correct but biologically meaningless2.
Why flow cytometry in drug development? Because better data builds better medicines
One thing is clear: flow cytometry is no longer just a research tool. It drives therapeutic insight. Its power has expanded even further with spectral flow cytometry, which enables high-dimensional, multiparameter analysis of cells, and with AI-driven data analysis, which extracts actionable insights from complex datasets.
Its importance continues to grow as therapeutic strategies become more complex and increasingly focus on modulating, engineering, or replacing cells rather than relying solely on small-molecule interventions. In this context, detailed cellular phenotyping and functional analysis are critical for understanding the mechanism of action and monitoring therapeutic effects. Today, it plays an increasingly critical role across the entire drug development pipeline, from early discovery and preclinical evaluation to clinical trials, manufacturing, and patient monitoring3.
For example, extracellular vesicles (EVs) are emerging as promising biomarkers and therapeutic platforms. Aibaidula et al. describe in the publication how spectral flow cytometry is leveraged for its ability to sensitively detect small particles while capturing the full emission spectrum of fluorochromes, enabling improved resolution in high-dimensional datasets. When combined with computational tools such as t-SNE and FlowSOM, this approach supports more robust population identification and reduces variability associated with manual gating4.
Flow cytometry as core technology
Taken together, these trends point to a clear conclusion: flow cytometry is becoming a core capability rather than a peripheral service. Advanced instruments, high-parameter assay design, and AI-supported data analysis are becoming standard expectations in modern clinical programs.
Of course, technology is only part of the story. Real impact comes from the people behind it. The future of flow cytometry belongs to organizations that can balance innovation with reliability, manage complexity without sacrificing clarity, and transform large, complex datasets into meaningful scientific and clinical insights.
Flow cytometry is growing more powerful, more complex, and more essential than ever. As technology accelerates, so does our responsibility to turn high‑dimensional data into clear, actionable insight. We’re excited to continue that journey!
In our next Flow Matters blog, we’ll bring these concepts to life through a therapeutic case study.
Key takeaways
- High‑parameter and spectral flow cytometry unlock deeper, more nuanced insight into cellular complexity, especially when working with limited or difficult sample types.
- AI and machine learning strengthen the entire flow cytometry workflow—from panel design to data interpretation—by improving consistency, scalability, and reproducibility.
- Advanced flow cytometry now plays a central role across the drug development pipeline, offering richer phenotyping, more sensitive detection, and stronger translational alignment.
- The future belongs to hybrid expertise: cutting‑edge tools paired with experienced scientific oversight, ensuring high‑dimensional data becomes meaningful, actionable insight.
References
- Konecny, A. J., Mage, P. L., Tyznik, A. J., Prlic, M. & Mair, F. OMIP‑102: 50‑color phenotyping of the human immune system with in‑depth assessment of T cells and dendritic cells. Cytometry Part A 105(6), 430–436 (2024). https://doi.org/10.1002/cyto.a.24841
- Ng, D. P., Simonson, P. D., Tárnok, A., Lucas, F., Kern, W., Rolf, N., Bogdanoski, G., Green, C., Brinkman, R. R. & Czechowska, K. Recommendations for using artificial intelligence in clinical flow cytometry. Cytometry Part B – Clinical Cytometry 106(4), 228–238 (2024). https://doi.org/10.1002/cyto.b.22166
- Ullas, S. & Sinclair, C. Applications of Flow Cytometry in Drug Discovery and Translational Research. International Journal of Molecular Sciences 25(7), 3851 (2024). https://doi:10.3390/ijms25073851
- Aibaidula, A., Fain, C. E., Cumba Garcia, L., Wier, A., Bouchal, S. M., Bauman, M. M., Jung, M. Y., Sarkaria, J. N., Johnson, A. J., & Parney, I. F. Spectral flow cytometry identifies distinct nonneoplastic plasma extracellular vesicle phenotype in glioblastoma patients. Neuro-Oncology Advances 2023;5(1):vdad082. https://doi.org/10.1093/noajnl/vdad082
About the Author
Amber Baele
Amber is an Associate Scientist on the Flow Cytometry team at Cerba Research, based in Ghent, Belgium. She joined the company in 2021 after earning her Master’s degree in Pharmaceutical Sciences – Drug Development from Ghent University. During her master’s thesis, she focused on instrument and method validation for a spectral flow cytometer at the clinical laboratory of Ghent University Hospital (UZ Ghent). In addition, she completed an internship at a community pharmacy, where she developed a strong patient-centered work ethic that continues to guide her scientific approach today.





