Matthias De Decker
Scientist, Flow Cytometry
Welcome to another edition of the Flow Matters blog! In our previous post, we explored the future of flow cytometry and the innovations shaping the field. Today, we’re diving into how flow cytometry is transforming virology and vaccine research, and how new technological advances are giving researchers an unprecedented view of the immune system in action.
Understanding how our immune system responds to viruses, and how vaccines can safely mimic and direct those responses, is central to developing safe and effective therapies. Over the past decades, flow cytometry has become one of the most powerful tools in immunology, virology, and vaccine research. By analyzing individual cells with incredible precision and speed, flow cytometry allows scientists to identify cell populations, measure functional responses, and detect molecular markers that were once impossible to measure. This level of detail is revolutionizing how we study infections and evaluate vaccines.
Decoding Virus-Host Interactions
Viruses are masters of survival. They invade host cells, hijack cellular machinery, and often evade immune detection in remarkably sophisticated ways. To counter this, scientists need tools that can dissect these interactions at a single cell level. This is where flow cytometry has become indispensable in virology research.
Today, flow cytometry provides a detailed window into the dynamic interplay between viruses and the immune system, enabling researchers to identify infected cells, characterize immune responses, and uncover mechanisms of immune evasion.
1. Identifying Infected Cells
A key application of flow cytometry in virology is the ability to distinguish infected from uninfected cells. By using fluorescently labeled antibodies targeting viral proteins, researchers can directly quantify infection at the cellular level.
- In HIV infected cells, detection of the viral p24 protein, a core structural component of the virus¹, allows precise measurement of infection rates and evaluation of antiviral therapies.
- During the COVID-19 pandemic, flow cytometry enabled identification of infected epithelial and immune cells through detection of the viral Nucleocapsid (N) protein². This approach supported real-time monitoring of SARS-CoV-2 replication and spread, while simultaneously profiling host immune responses.
2. Characterizing Immune Responses
Understanding how the immune system reacts to viral infection is essential for therapeutic and vaccine development. Flow cytometry excels at breaking down these responses into measurable components.
- Identification of key immune cell populations, including CD4⁺ helper T cells, CD8⁺ cytotoxic T cells, B cells, and Natural killer (NK) cells.
- Assessment of activation, proliferation, and exhaustion of immune cells through detection of specific markers such as CD28, CD38, CD25, HLA-DR, Ki-67, TIM-3, and LAG-3.
- Functional profiling via intracellular cytokine staining (e.g., IFN-γ and IL-2), revealing the effectiveness of antiviral responses (Figure 1).
By tracking these parameters over time, researchers can identify which immune mechanisms correlate with viral clearance and protection.
3. Studying Virus-Host Interactions
Viruses do more than just infect cells; they actively reshape them. Some viruses downregulate surface molecules to “hide” from immune cells, while others manipulate signaling pathways to suppress host defenses.
Flow cytometry makes these subtle yet critical changes visible. It can reveal:
- Alterations in receptor expression that influence viral entry.
- Induction of apoptosis (programmed cell death).
- Modulation of intracellular signaling pathways involved in immune activation or suppression.
These insights are crucial for understanding viral strategies of immune evasion. By uncovering these mechanisms, flow cytometry helps guide the development of antiviral therapies, not just by targeting the virus itself, but by restoring or enhancing the host immune response.
A Window into Immune Protection
Vaccines are designed to train the immune system to recognize and respond to pathogens, arming the body against future infections. To evaluate how well a vaccine works, scientists need to look beyond simple antibody responses and assess the full spectrum of immune activity, including antigen-specific T cell responses. Flow cytometry has become an indispensable tool for this purpose, offering detailed insights into both cellular and humoral (antibody-mediated) immunity.
1. Measuring T Cell Responses
T cells are central to antiviral immunity, particularly when antibody responses are insufficient or decline over time. Flow cytometry enables researchers to examine T cell responses with remarkable precision³:
- Antigen-specific T cells: Using peptide-MHC tetramers, scientists can identify T cells that specifically recognize viral epitopes, providing a direct measure of vaccine-induced cellular immunity.
- Functional activity: Cytokine production (such as IFN-γ and TNF-α; Figure 1) and degranulation markers like CD107a in CD8⁺ cytotoxic T cells or NK cells reveal how effectively these cells can respond to infected targets.
- Phenotypic profiling: Deep immunophenotyping to identify subsets of antigen-specific T cells, such as naïve, central memory, effector memory, and terminally differentiated T cells. Identifying subsets help to map the durability and quality of the vaccine-induced immune response. In addition, activation, proliferation and exhaustion markers further inform on the functional state of these immune cells.
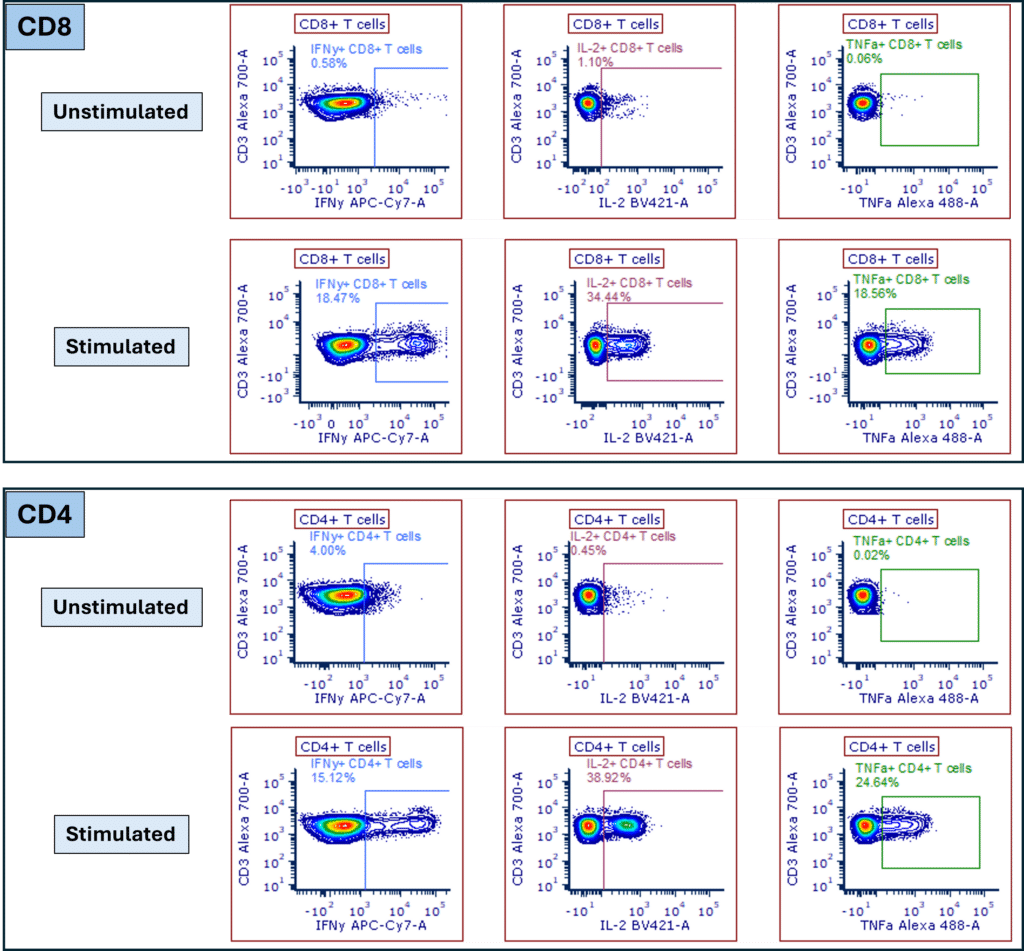
Figure 1: Intracellular cytokine staining reveals T cell activation. Detection of IFN-γ, IL-2 and TNF-α in CD8+ (top) and CD4+ (bottom) T cell subsets in PBMCs stimulated with PMA and Ionomycin, compared to unstimulated PBMC controls. Cells were gated on singlets, live cells and CD3+ lymphocytes.
2. Evaluating B Cell and Antibody Responses
Flow cytometry can provide a detailed view of B cell and antibody-mediated immunity⁴, complementing T cell analyses:
- Antigen-specific B cells: Fluorescently labeled viral proteins allow researchers to track B cells that recognize the target pathogen
- Class switching and maturation: Monitoring markers like IgM, IgG, and IgA reveals how B cells evolve in response to vaccination, indicating the development of high-affinity, protective antibodies
- Plasma cells: Quantifying these antibody-secreting cells offers insight into the magnitude of the humoral response and the potential longevity of vaccine-induced protection
Together, the detailed characterization of antigen-specific T cells alongside parallel assessment of antigen-specific B cells and plasma cell responses, provides a comprehensive, single cell-level view of vaccine-induced immunity.
This integrated approach links T cell functionality with humoral responses to better define the overall effectiveness and durability of protection, while highlighting the critical role of T cell responses, particularly for vaccines against viruses such as HIV, influenza, and SARS-CoV-2, where robust cellular immunity can be a key determinant of protection (Figure 2).
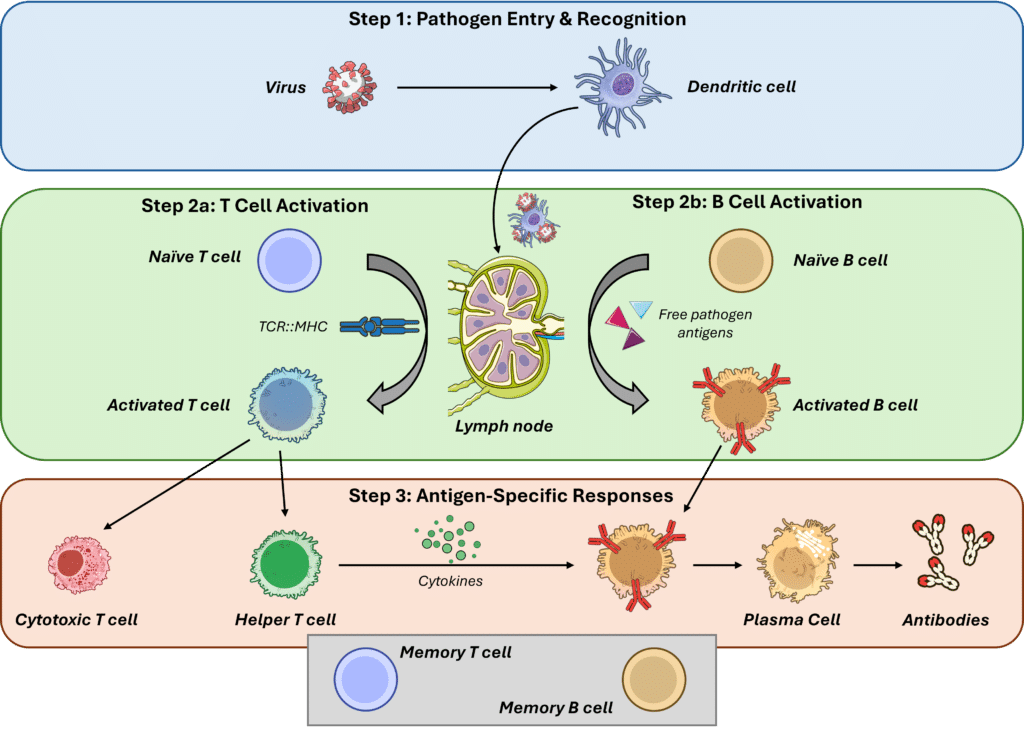
Figure 2: Activation of the immune system during viral infection leads to the generation of antigen-specific T cells, B cells, and plasma cells, thereby connecting T cell functionality with humoral immune responses.
Inside the Immune Response: Measuring Influenza Peptide Activity with Flow Cytometry
Imagine you have just received your seasonal flu vaccine. Within days, your immune system is already at work, but how do scientists actually see that response happening?
This is where flow cytometry comes in.
In a real-world clinical study, participants receive the influenza vaccine, which contains viral peptides that “teach” the immune system to recognize the virus. After vaccination, researchers collect small blood samples at specific time points to monitor how the immune system reacts. Using flow cytometry, they can pinpoint exactly which cells respond. With tools like peptide-MHC tetramers, influenza-specific T cells can be identified with high precision, while other key immune cells, such as B cells and NK cells, are analyzed at the same time, providing a more complete picture of the immune response.
But it does not stop there.
At the same time, researchers can assess what those antigen‑specific cells are doing functionally. For example, are they producing key signaling cytokines such as IFN‑γ or TNF‑α? These molecules play a critical role in coordinating immune responses by activating other immune cells and enhancing antiviral defenses, thereby supporting effective pathogen control.
The result? A detailed snapshot of how the body reacts to the vaccine. Studies using this approach have shown that people who generate stronger, multifunctional T cell responses often develop better protection against influenza
Seeing More, Knowing More: Spectral Flow Shapes Vaccine Research
Flow cytometry has come a long way from its traditional roots, evolving into a powerful, high-dimensional tool for immune profiling. Among the most exciting advancements is spectral flow cytometry, a technology that is redefining how scientists study immune responses. By enabling high-parameter analysis, spectral flow cytometry allows researchers to explore the complexity of vaccine-induced immunity in ways that were not possible with conventional approaches.
So, what makes it such a game changer?
First, spectral flow cytometry dramatically increases the number of markers that can be measured simultaneously while minimizing issues caused by spectral overlap. This not only expands the depth of analysis but also simplifies panel design and reduces the need for complex compensation strategies.
With this expanded parameter space, researchers can move beyond fragmented analyses and instead capture a holistic picture of immune responses in a single experiment. Peptide-MHC tetramers enable detailed profiling of antigen-specific T cells, while B cells and plasma cell responses can be evaluated in parallel. This integrated approach preserves biological context and eliminates the variability associated with splitting samples across multiple panels, as all markers are combined in a single tube to enable simultaneous assessment of multiple cell populations and their multifunctionality.
Another major advantage is the ability to identify rare but critical immune cell populations. These cells, often overlooked in lower-dimensional analyses, can play an essential role in protective immunity, especially for next-generation vaccines targeting complex or rapidly evolving pathogens. In such cases, subtle immune signatures may be the key to understanding why a vaccine succeeds or fails.
Ultimately, spectral flow cytometry is helping shift vaccine research from a reductionist approach to a systems-level understanding of immunity. By enabling comprehensive, high-dimensional immune profiling, it is improving the discovery of meaningful biomarkers and accelerating vaccine development with more predictive insights into immunogenicity and long-term protection.
And as this technology continues to evolve alongside innovations like imaging cytometry, integrating quantitative single cell analysis with high-resolution imaging, and single cell multiomics approaches such as CITE-Seq5, which combine protein and gene expression profiling at the single cell level, the field is becoming increasingly powerful and highly informative.
Improvements in AI-driven analysis could further facilitate faster and more accurate interpretation of highly complex, multiparametric panels, making the future of vaccine research more precise, more integrated, and more promising than ever.
References
- Tessema B, Boldt A, König B, Maier M, Sack U. Flow-Cytometry Intracellular Detection and Quantification of HIV1 p24 Antigen and Immunocheckpoint Molecules in T Cells among HIV/AIDS Patients. HIV AIDS (Auckl). 2022 Aug 4;14:365-379. doi: 10.2147/HIV.S374369. PMID: 35958525; PMCID: PMC9359413.
- Vinit Upasani, Marjolein Knoester, Daniele Pantano, Lili Gard, Jolanda M. Smit, Bernardina T.F. van der Gun, Adriana Tami, Izabela A. Rodenhuis-Zybert. Evaluation of a flow cytometry-based surrogate assay (FlowSA) for the detection of SARS-CoV-2 in clinical samples. Journal of Clinical Virology Plus, Volume 5, Issue 1, 2025, 100204, ISSN 2667-0380, https://doi.org/10.1016/j.jcvp.2025.100204.
- Newell EW, Davis MM. Beyond model antigens: high-dimensional methods for the analysis of antigen-specific T cells. Nat Biotechnol. 2014 Feb;32(2):149-57. doi: 10.1038/nbt.2783. Epub 2014 Jan 19. PMID: 24441473; PMCID: PMC4001742.
- Broketa M, Bruhns P. Single-Cell Technologies for the Study of Antibody-Secreting Cells. Front Immunol. 2022 Jan 31;12:821729. doi: 10.3389/fimmu.2021.821729. PMID: 35173713; PMCID: PMC8841722.
- Stoeckius, M., Hafemeister, C., Stephenson, W. et al.Simultaneous epitope and transcriptome measurement in single cells. Nat Methods 14, 865–868 (2017). https://doi.org/10.1038/nmeth.4380
About the Author
Matthias De Decker
Matthias is a Senior Scientist on the Flow Cytometry team at Cerba Research, based in Ghent, Belgium. He joined the company in 2021 after earning his PhD in Immunology and a master’s degree in biology from Ghent University. His expertise lies in human hematopoiesis, with a particular focus on normal T cell development.





